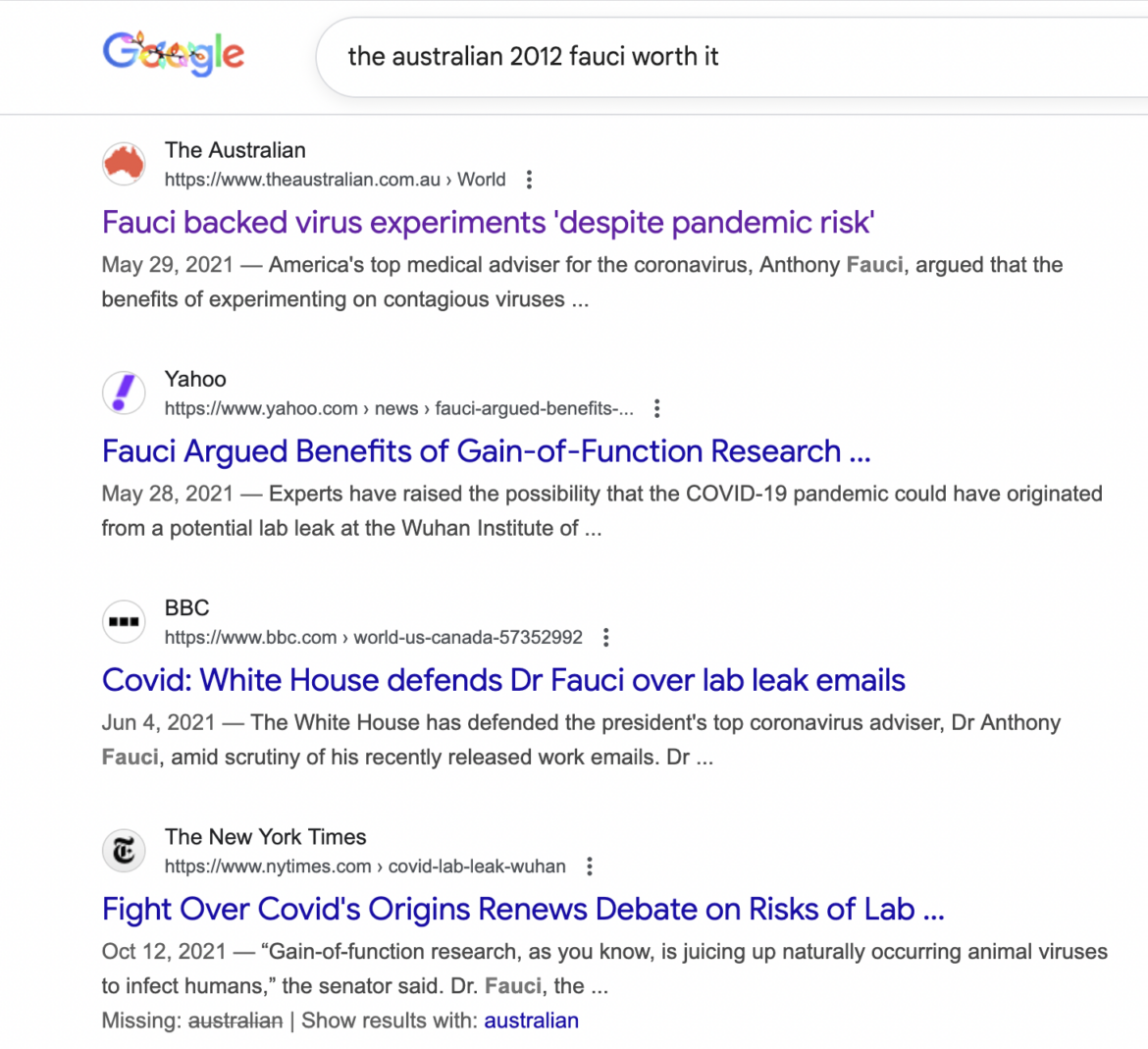
Yesterday, the Department of Justice indicted David Morens—a longtime senior advisor to Anthony Fauci—on charges including conspiracy and destroying federal records.
Translation? The COVID narrative wasn’t just “messy science.” It was engineered.
In this episode of The Independents, I break down how “settled science” was manufactured—from the Peter Daszak-orchestrated Lancet letter to the suppression of the lab-leak hypothesis, the targeting of dissenting scientists, and the media amplification that made questioning any of it seem insane.
I was talking about the lab leak in early 2020. I got censored, shadowbanned, and labeled a conspiracy theorist.
Now? There are indictments.
We walk through:
- How the narrative flipped behind closed doors
- The emails, the funding, and the conflicts of interest
- Why dissenting voices were crushed
- And what this means for trust, science, and you
If you felt gaslit during Covidmania—you weren’t crazy.
You were early.
👉 Like, share, and subscribe if you believe truth still matters.
REMINDER: “Gain-of-Function” means taxpayers paid to: “enhance traits like transmissibility, virulence, immune evasion, or other pandemic-relevant properties” of a virus. Covidmania was your government screwing you over from start to finish. Are you mad yet?
Nope, many of you are still ensnared in the Left/Right paradigm, and are not ready for the truth…
The time frame in Fauci’s pardon is a tell, too. Covers from 2014. Why’s that? Because that’s when they started funding Bat Corona Virus work (you know: GAIN OF FUNCTION).
Gain-of-function (GoF) research is a type of scientific study—typically in virology, microbiology, or related fields—that genetically modifies an organism (most often a virus, bacterium, or other pathogen) to enhance or add new biological functions or traits. These enhancements can include increased transmissibility (ability to spread), virulence (ability to cause disease), pathogenicity, host range (ability to infect new species or cell types), immune evasion, or replication efficiency.
The goal is usually to better understand how pathogens evolve in nature, predict and prepare for potential pandemics, identify drug/vaccine targets, or study virus-host interactions. It is a subset of “dual-use research of concern” because the same techniques that advance medicine can also create more dangerous pathogens (intentionally or accidentally). GoF is the opposite of “loss-of-function” research, which disables or reduces traits. Not all GoF is high-risk; the term is broad, and only specific “GoF of concern” or “potential pandemic pathogen” (PPP) experiments (those reasonably expected to make influenza, SARS-like, or MERS-like viruses more dangerous to mammals via respiratory routes) have triggered U.S. policy oversight, such as the 2014–2017 funding pause.
U.S.-Funded Bat Coronavirus Research (2014–Present) as a Prominent Example
A major real-world case involves U.S. National Institutes of Health (NIH) funding to the nonprofit EcoHealth Alliance (led by Peter Daszak) for work on bat coronaviruses, primarily in collaboration with the Wuhan Institute of Virology (WIV) in China. This illustrates GoF-type experiments in practice and the intense controversy around them.
- 2014 Grant Award and Context: In June 2014, NIH awarded EcoHealth Alliance a multi-year grant (R01AI110964, “Understanding the Risk of Bat Coronavirus Emergence,” ~$3.7–4+ million total through extensions) to study how bat coronaviruses in China might spill over to humans. EcoHealth sub-awarded roughly $600,000+ to WIV for fieldwork (bat sampling and sequencing) and lab experiments. This overlapped with the October 2014 U.S. government pause on federal funding for certain GoF research on influenza, SARS, and MERS viruses (due to biosafety concerns after lab accidents). The EcoHealth grant was already in place or grandfathered and continued, with NIH later adding reporting conditions in 2016 out of caution.
- What the Research Involved: Scientists collected bat samples, sequenced novel coronaviruses, and performed genetic engineering to create chimeric (hybrid) viruses. For example, they took the backbone of a known bat coronavirus (WIV1) and swapped in spike proteins (the surface protein that determines cell entry) from other recently discovered bat coronaviruses. These chimeras were then tested in cell cultures and humanized mice (mice engineered with human ACE2 receptors, the entry point for SARS-like viruses) to assess binding to human cells, replication efficiency, and disease potential. Some experiments showed enhanced abilities—e.g., certain modified viruses could infect human cells or grow far more efficiently in mice than the parental strains. Related 2015 work (involving EcoHealth collaborators like Ralph Baric) produced a chimeric virus capable of replicating in human airway cells.
- 2018 DEFUSE Proposal (Unfunded but Relevant): EcoHealth, with Baric and WIV, submitted a proposal to DARPA called DEFUSE. It outlined more ambitious engineering: creating full-length infectious clones of bat SARS-related coronaviruses, inserting proteolytic cleavage sites (including furin cleavage sites that help viruses enter human cells), and testing in humanized models. DARPA rejected it, citing risks. Some elements echoed ongoing NIH-funded work.
- 2020–2024 Developments and Controversy: The grant was suspended in April 2020 (early in the COVID-19 pandemic) amid questions about WIV’s role in the outbreak. It was partially reinstated with stricter conditions but faced repeated scrutiny. In 2021, NIH Principal Deputy Director Lawrence Tabak informed Congress that EcoHealth failed to promptly report that some chimeric viruses had become ~10,000 times more infectious in humanized mice—results that triggered grant reporting requirements. Tabak and later testimony clarified that NIH had funded experiments meeting a common-sense definition of GoF (enhancing viral properties), though officials maintained it did not violate the exact 2014 pause criteria or P3CO oversight framework at the time. EcoHealth disputed characterizations and said the work was for risk assessment. In 2024, the Department of Health and Human Services (HHS) suspended all EcoHealth grants and debarred the organization for years, citing compliance failures, inadequate oversight of WIV, and biosafety risks.
- Key Debate and Outcomes: Proponents argue this research was essential for pandemic preparedness and that the viruses studied were genetically too distant from SARS-CoV-2 to have caused the pandemic. Critics (including some lawmakers and scientists) contend it constituted GoF of concern, posed lab-leak risks, and violated the spirit (if not letter) of funding restrictions—especially given WIV’s biosafety level and China’s track record. NIH has emphasized no evidence links this specific work to SARS-CoV-2 origins, and oversight has since tightened. The episode fueled broader policy changes on GoF/P3CO reviews.
In summary, this bat coronavirus research exemplifies GoF because it deliberately created novel viral variants with potentially improved human infectivity to study emergence risks. It highlights the trade-offs: valuable for science but carrying biosafety/biosecurity concerns that led to pauses, reporting failures, and eventual funding cuts. U.S. policy continues to evolve to balance benefits and risks.
List all the “GoF of concern” or “potential pandemic pathogen” (PPP) experiments the US is currently funding
As of May 2026, there is no publicly available comprehensive list of specific “Gain-of-Function (GoF) of concern” or “Potential Pandemic Pathogen (PPP / PEPP)” experiments currently being funded by the U.S. government.
Current U.S. Policy Context (Post-May 2025)
In May 2025, President Trump issued Executive Order 14292 (“Improving the Safety and Security of Biological Research”). It directed federal agencies to:
- Immediately end funding for “dangerous gain-of-function research” conducted by foreign entities (especially in countries of concern like China) or in nations lacking adequate U.S.-equivalent oversight.
- Suspend other dangerous GoF research domestically until a new, stricter policy is developed.
The National Institutes of Health (NIH) responded with implementation notices:
- NOT-OD-25-112 (May 2025) and NOT-OD-25-127 (June 2025): These required awardees to review their portfolios for dangerous GoF research, halted new applications/proposals after May 7, 2025, and mandated suspension or termination of ongoing projects meeting the definition (broadly covering research on infectious agents/toxins that could enhance pandemic potential or pose significant biosafety/biosecurity risks).
This effectively paused the 2024 U.S. Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential (DURC-PEPP), which had been scheduled to take effect May 6, 2025. A replacement policy is under development, but high-risk research remains restricted in the interim.
As a result:
- Dozens of projects (reports cite ~40+ flagged, with broader portfolio reviews covering hundreds) were reviewed for suspension/termination.
- No new federal funding for dangerous GoF/PPP research is being accepted.
- Existing high-risk work (if any) is under suspension or has had funds redirected/terminated.
Historical Context (Pre-2025 Under HHS P3CO Framework)
Prior to the 2025 EO, the main oversight mechanism for PPP research was the 2017 HHS P3CO Framework (for “enhanced Potential Pandemic Pathogens”). This applied only to a narrow subset of experiments reasonably anticipated to create, transfer, or use enhanced pathogens with significant pandemic potential.
Public records show extremely limited activity:
- Only a handful of projects (~3–4 total) were ever formally referred to the HHS P3CO Review Group for extra scrutiny.
- Examples (all influenza-related, via NIH RePORTER):
- Two projects (e.g., studies on H7N9 and avian influenza transmissibility in mammals) were approved with mitigations but later ended (one in 2020, support for others concluded by 2021).
- Others (including a 2019 Centers of Excellence for Influenza Research and Surveillance component and a 2024 proposal) had their ePPP/GoF portions not funded, removed, or redirected to lower-risk alternatives.
- No broad or ongoing slate of active PPP/GoF-of-concern experiments was ever publicly detailed beyond these.
Why No Full Public List Exists
- Biosecurity and national security considerations limit detailed public disclosure of specific high-risk projects.
- Oversight (via Institutional Review Entities, agency reviews, and now the EO) occurs case-by-case rather than through a centralized public registry.
- A March 2026 GAO report criticized HHS for insufficient transparency on high-risk pathogen research oversight but did not identify or list any specific ongoing funded experiments.
In summary: Due to the 2025 Executive Order and NIH actions, the U.S. is not actively funding new (or, in many cases, continuing) GoF-of-concern or PPP experiments as previously defined. Any pre-existing work meeting the “dangerous” criteria has been suspended, terminated, or had risky components removed. If you need details on a specific grant, pathogen, or agency, provide more context and I can search further. Policy in this area continues to evolve.
Find everything related to “(DURC-PEPP)” programs
DURC-PEPP refers to the United States Government (USG) Policy for Oversight of Dual Use Research of Concern (DURC) and Pathogens with Enhanced Pandemic Potential (PEPP), a 2024 federal oversight framework for certain high-risk life sciences research. It is not a set of dedicated funding “programs” or grant initiatives. Instead, it is a regulatory policy that applies additional review, risk assessment, and mitigation requirements to qualifying federally funded research involving biological agents, toxins, or pathogens that could pose biosafety, biosecurity, or dual-use risks (i.e., beneficial science that could also be misused to cause harm).
The policy was never fully implemented due to a May 2025 Executive Order and remains in a paused/superseded state as of May 2026. Below is a complete summary of everything related to it, based on official sources.
1. Background and History
- Predecessors:
- 2012/2014 USG DURC Policies: Focused on a narrow list of ~15 agents/toxins and specific experimental outcomes with clear misuse potential.
- 2017 HHS P3CO Framework (and OSTP guidance): Addressed “enhanced potential pandemic pathogens” (ePPPs) with pandemic potential.
- 2024 DURC-PEPP Policy: Released by the White House Office of Science and Technology Policy (OSTP) on May 6, 2024. It unified and expanded the prior frameworks into one comprehensive policy to better address evolving risks while supporting beneficial research.
- Effective Date: Originally scheduled for May 6, 2025, applying to all federally funded intramural/extramural research (grants, contracts, cooperative agreements, etc., across agencies like NIH, HHS, USDA, etc.).
2. Scope and Key Definitions (2024 Policy)
The policy covers research reasonably anticipated to involve:
- Category 1 (DURC): Research with one or more of a specified list of biological agents/toxins (expanded significantly from prior policies, reportedly to ~91 agents/toxins) that is anticipated to produce one of several listed experimental outcomes (e.g., effects on transmissibility, virulence, immune evasion, host range, etc.) and meets a DURC risk assessment.
- Category 2 (PEPP): Research involving pathogens with enhanced pandemic potential—i.e., work on (or creating) pathogens likely capable of wide human spread and moderate-to-severe disease, where experiments enhance traits like transmissibility, virulence, immune evasion, or other pandemic-relevant properties.
Purpose: Balance scientific benefits (e.g., pandemic preparedness) with minimizing risks of misuse, accidental release, or proliferation. It does not replace Select Agent regulations or other biosafety rules but complements them.
3. Oversight Requirements and Process
- Principal Investigator (PI) Responsibilities: Self-assess proposed/ongoing research against Category 1/2 criteria at proposal stage and throughout the project; report to the institution and funding agency.
- Institutional Review Entity (IRE): Usually the Institutional Biosafety Committee (IBC) or equivalent. Conducts case-by-case review, risk-benefit analysis, and approves mitigation plans.
- Funding Agency Role: Additional federal-level review, approval of mitigation plans, and ongoing oversight. May impose conditions, pause funding, or require modifications.
- Key Steps: Identification → IRE review → Risk mitigation plan → Federal notification/reporting → Compliance monitoring. Noncompliance can lead to funding suspension/termination.
- Implementation Guidance (issued alongside the policy): Detailed FAQs, examples, roles, and processes for institutions and agencies.
The policy encouraged (but did not require) similar oversight for non-federally funded research.
4. Related Documents and Resources
- Main Policy PDF (May 2024): https://aspr.hhs.gov/S3/Documents/USG-Policy-for-Oversight-of-DURC-and-PEPP-May2024-508.pdf
- Implementation Guidance PDF (May 2024): https://aspr.hhs.gov/S3/Documents/USG-DURC-PEPP-Implementation-Guidance-May2024-508.pdf (or archived White House versions)
- NIH Implementation Notice (rescinded): NOT-OD-25-061 (Jan 10, 2025) — outlined NIH-specific rollout.
- HHS/ASP R pages: Dual Use Research of Concern Oversight Policy Framework and history.
Most major research universities (e.g., Michigan, Harvard, Duke, Florida, etc.) issued internal DURC-PEPP compliance policies mirroring the federal one.
5. 2025 Executive Order and Current Status (as of May 2026)
- Executive Order 14292 (“Improving the Safety and Security of Biological Research”), issued May 5, 2025, by President Trump:
- Immediately paused/suspended federal funding for “dangerous gain-of-function research” (broadly defined as work on infectious agents/toxins that enhances pathogenicity, transmissibility, immune evasion, host range, etc., with significant societal consequences).
- Directed OSTP to revise or replace the 2024 DURC-PEPP Policy within 120 days (by ~September 2025) with stronger oversight, clearer scope, enforcement, audits, and transparency.
- Ended funding for such research in foreign entities (especially “countries of concern” like China) or jurisdictions lacking U.S.-equivalent oversight.
- Required new enforcement terms in grants/contracts and strategies for non-federal research.
- NIH Response (NOT-OD-25-112, May 7, 2025): Rescinded prior implementation notice; halted new applications for dangerous GoF; suspended ongoing funding per forthcoming guidance. Awardees must proactively review and halt qualifying work. A June 2025 update addressed terminations/suspensions.
- Current Status: The 2024 DURC-PEPP Policy was superseded before full rollout. A replacement policy has not been publicly released as of May 2026 (beyond the 120-day deadline). High-risk (“dangerous GoF”) research remains paused/suspended. Institutions continue to apply prior DURC/P3CO rules where relevant and await new federal guidance. No new federal funding is being accepted for covered high-risk work.
6. Funded Research / “Programs” Under DURC-PEPP
- There are no dedicated DURC-PEPP “programs” or a public centralized list of projects. The framework applies oversight to any qualifying federally funded research on a case-by-case basis.
- Under the narrower predecessor P3CO framework, only a handful of projects (mostly influenza-related) were ever formally reviewed; most high-risk proposals were modified, not funded, or redirected.
- No specific ongoing or historical DURC-PEPP-funded experiments have been publicly detailed due to biosecurity sensitivities. Portfolio reviews occurred, but details remain non-public.
In summary, DURC-PEPP was a short-lived (pre-implementation) expansion of U.S. biosecurity oversight for dual-use and pandemic-potential pathogen research. It was overtaken by the 2025 EO prioritizing stricter controls and pauses on dangerous gain-of-function work. For the most current status or institution-specific compliance, check NIH/HHS guidance or your organization’s research compliance office. Policy development in this area continues to evolve.